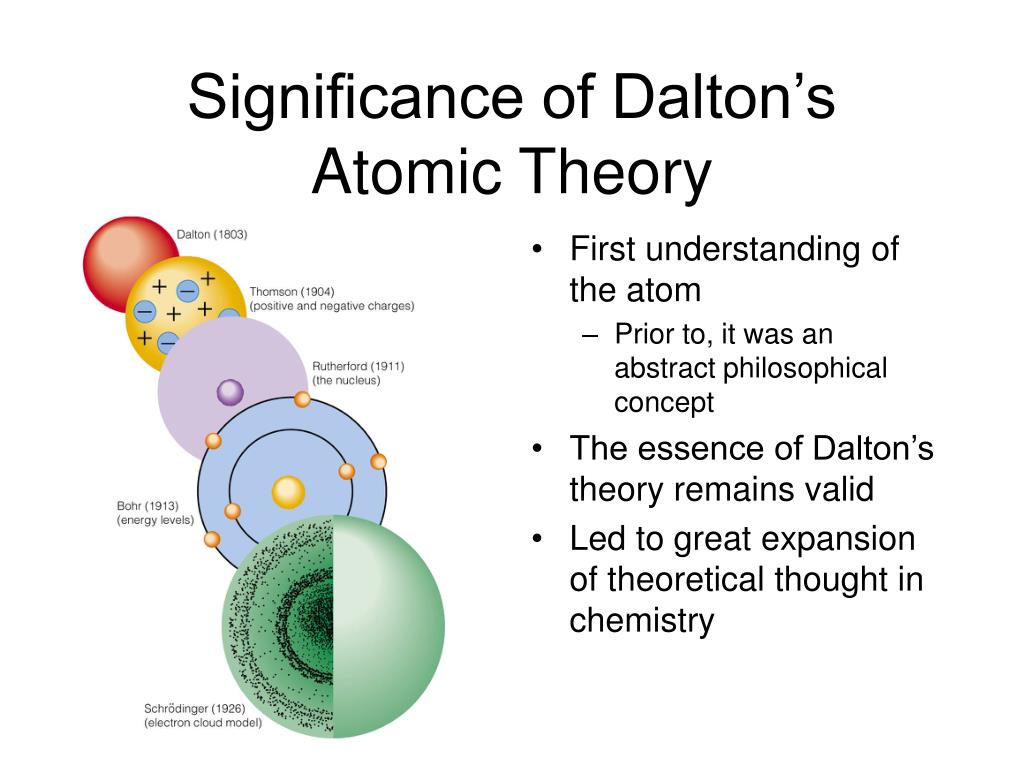 
So, different substances necessarily have different boiling points, melting points, and other properties. For example, the atoms of zinc are different from the atoms of gold, etc. He also explained that the atoms of one element are different from the atoms of other elements. For example, all the atoms present in zinc are the same. Explanation of Postulatesĭalton worked and expanded the work of Leucippus of Miletus (5 th-century B.C.E) who assumed that all substances are made up of solid, hard, impenetrable, and mobile particles, which he called “ atomos” (literally indivisible) in around 430 B.C.E.ĭalton proposed that every single atom of an element is the same as other atoms of that element. They can always be recovered, and never change in a chemical reaction. Atoms can neither be created nor destroyed.Chemical compounds are formed by the combination of atoms of different elements in specific ratios.Atoms of different elements are different in terms of their atomic weights and chemical properties.Atoms of a single element are always identical.Each element is made up of tiny and indestructible particles called atoms.There are five basic postulates of Dalton’s atomic theory. Postulates of John Dalton’s Atomic Theory 
However, the law of constant compositions stated that pure substances or compounds have always the same composition of their elements. The law of conservation of mass, which was devised by Antoine Lavoisier in 1789, stated that matter is neither created nor destroyed in a closed system. This time frame exists in both the 17 th and 18 th centuries when John Dalton carried on the work started by Robert Boyle and Antoine Lavoisier and took it to the development of his atomic theory.ĭalton’s atomic theory is based on the law of conservation of mass and the law of constant compositions. The works of Robert Boyle, Antoine Lavoisier, Issac Newton, John Dalton, and Joseph Priestly paved the path for chemistry to be considered a proper science. “In order to convey a knowledge of chemical facts and experience, the more clearly, it has been generally deemed best, to begin with, the description of such principles or bodies as are the most simple, then to proceed to those that are compounded of two simple elements, and afterward to those compounded of three or more simple elements….” On the elemental (atomic) composition of compounds, he says that In his book, A New System of Chemical Philosophy, he explained his concepts of heat, specific heat, expansion by heat, elastic fluids, the constitution of bodies and their chemical synthesis, etc. Although, his atomic theory had some shortcomings afterward, still, it was the first-ever attempt to completely describe matter in terms of atoms. In the year 1808, he introduced a scientific theory on the nature of matter. Furthermore, he also worked for color blindness, which is today known as Daltonism, in his honor. One of his biggest achievements is the introduction of atomic theory. John Dalton (1766-1844) was an English physicist, meteorologist, and a well-known chemist of his time.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
